Fundamentals of Clinical Research
Fundamentals of Clinical Research is a practical training program that provides a clear understanding of how clinical trials are conducted at the operational and site level. The course is designed to introduce core clinical research concepts and workflows that support consistent execution and trial readiness. The program focuses on real-world application rather than theory, giving participants a structured foundation in clinical trial operations.
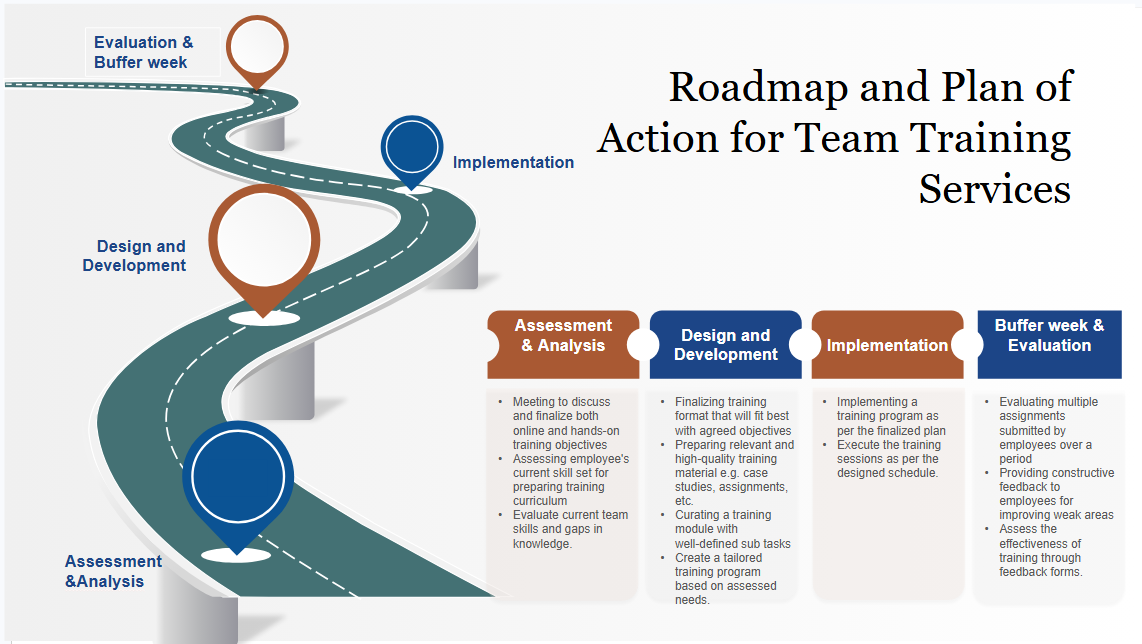
Learning Objectives:
Upon completion of the CEC Fundamentals Program, participants will be able to:
- Apply GCP principles and regulatory standards to ensure compliant, high- quality clinical trial execution
- Execute core CRC responsibilities aligned with site operations, sponsor expectations, and study protocols
- Demonstrate job-ready competencies, professionalism, and decision- making required to support effective study coordination
Program Features & Specifications:
- Expert-led training delivered by experienced clinical research professionals
- Blended learning model combining self-paced modules with instructor- led sessions
- Multi-format content (text, audio, hands-on) to support diverse learning styles
- Cohort-based structure to encourage collaboration and engagement
- Flexible delivery options (in-person, virtual, and train-the-trainer)
- Structured assessment to validate learning outcomes and demonstrate impact
Program Structure:
- Program Overview & Learning Roadmap
- Clinical Research Foundations: Program Orientation
Core Modules
Introduction to Clinical Research
This module provides a structured overview of the clinical research landscape, including key stakeholders, regulatory frameworks, and the full study lifecycle. Participants will gain a clear understanding of how trials are designed, executed, and managed across different phases. It sets the foundation for building practical, real-world competency in clinical operations.
Informed Consent
This module focuses on the ethical and regulatory requirements of the informed consent process, emphasizing patient safety and autonomy. It provides practical guidance on conducting consent discussions, ensuring comprehension, and maintaining proper documentation. Learners will understand how to handle real-world challenges while remaining compliant.
ALCOA+
This module introduces the principles of ALCOA+ and their critical role in maintaining data integrity in clinical trials. It goes beyond definitions to demonstrate how these standards are applied in daily documentation and data handling. Participants will learn how to ensure data is audit-ready and defensible at all times.
How to Read a Protocol
This module teaches a systematic approach to reading and interpreting clinical trial protocols. It highlights key sections, critical endpoints, and operational requirements that impact site execution. Participants will develop the ability to translate protocol content into practical actions within a study setting.
Phases of Clinical Trial
This module provides a comprehensive understanding of each phase of clinical development, from early safety studies to post-marketing surveillance. It explains the objectives, design differences, and regulatory expectations across phases. Learners will gain clarity on how each phase contributes to overall drug development.
Source Documentation
Types of Sponsor/CRO Visit
This module outlines the different types of sponsor and CRO visits, including site initiation, monitoring, close-out, and audits. It explains the purpose, expectations, and preparation strategies for each type. Learners will gain confidence in managing these interactions effectively and professionally.
Investigational Product
Adverse Events and Types
Protocol Deviations / CAPA
This module explains how to identify, document, and assess protocol deviations and their impact on study integrity. It also introduces the principles of corrective and preventive actions (CAPA) to address root causes. Participants will learn how to implement effective solutions and prevent recurrence.
Importance of QA/QC with Implementation
This module explores the role of quality assurance and quality control in ensuring consistent and compliant clinical trial execution. It provides practical strategies for implementing QA/QC processes within daily operations. Learners will understand how to build a culture of quality and continuous improvement.
Subject Recruitment and Retention
This module focuses on practical and ethical strategies to enhance patient recruitment and retention in clinical trials. It addresses common challenges and provides actionable solutions to improve enrollment outcomes. Participants will learn how to balance efficiency with patient-centered approaches.
Risk Assessment and Management in Clinical Trial
This module introduces risk-based approaches to clinical trial management, focusing on identifying, evaluating, and mitigating potential risks. It highlights the importance of proactive planning and continuous monitoring. Learners will gain tools to improve study efficiency and reduce operational issues.
FDA Findings During Audit
This module provides insight into common FDA audit findings and areas of concern in clinical trials. It emphasizes proactive preparation, documentation practices, and inspection readiness. Participants will learn how to respond effectively and maintain compliance under scrutiny.
Regulatory Documents
This module provides insight into common FDA audit findings and areas of concern in clinical trials. It emphasizes proactive preparation, documentation practices, and inspection readiness. Participants will learn how to respond effectively and maintain compliance under scrutiny.
Procedures and Equipment Tutorials
This module offers practical guidance on performing key clinical procedures and using study-related equipment correctly. It focuses on consistency, accuracy, and adherence to protocol requirements. Participants will develop confidence in executing procedures in real-world settings.
Contract and Budget
This module provides an overview of clinical trial budgeting and contract fundamentals. It covers cost considerations, negotiation strategies, and financial planning. Learners will understand how to align operational needs with financial feasibility.
SOPs
This module explains the importance of standard operating procedures in maintaining consistency and compliance. It provides guidance on developing, implementing, and following SOPs effectively. Participants will learn how SOPs support quality, efficiency, and regulatory readiness.
Program Conclusion
Training Reflections, Key Takeaways, and Program Wrap-Up
** Training programs are offered separately from consulting engagements


